Immune suppressing treatments - an essential component of many patient's cancer care - can blunt vaccine immune response
Rye Brook, N.Y., September 13, 2021 – More than one in two patients with B-cell blood cancers produced antibodies to a third dose of COVID-19 vaccine despite having no detectable antibodies after the first two doses, according to a new study from The Leukemia & Lymphoma Society (LLS). The study also showed that patients who had detectable antibodies after the first two vaccine doses had increased levels after the third dose. The study was published this week in the journal Cancer Cell (see full study and tables).
“The additional COVID-19 vaccine dose appears to be improving immune response in many people with blood cancer—one of many conditions that can suppress a person’s immune system,” said Dr. Gwen Nichols, Chief Medical Officer at LLS. “However, while vaccination offers protection to the majority of blood cancer patients, some will not mount a full antibody response even with this additional dose.”
Get vaccinated, continue to take preventive precautions
LLS recommends that blood cancer patients who received two doses of either Moderna or Pfizer mRNA vaccine get an additional dose according to the Centers for Disease Control and Prevention (CDC) guidance for people who are moderately to severely immunocompromised. CDC has indicated that additional guidance will be forthcoming for patients who received the Johnson & Johnson’s Janssen COVID-19 vaccine. Until then, patients who received J&J or any other non-mRNA vaccine should consider an additional dose and talk to their healthcare provider.
LLS also advises all blood cancer patients to continue to layer on additional precautions like masking and social distancing to avoid becoming infected. Based on pre-vaccine data, patients with blood cancer tend to have more prolonged and severe COVID-19 infections compared to their healthy counterparts, including having significantly higher rates of death.
Study provides insights about the effect of immune suppressing treatments on immune response
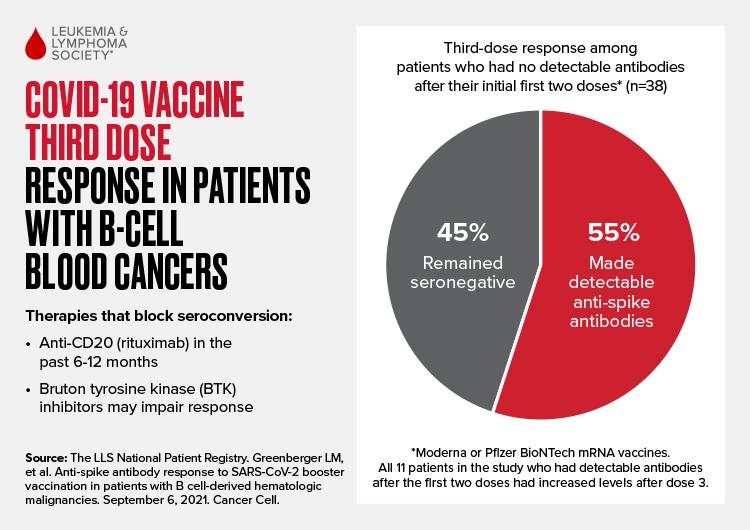
Treatment with the antibody rituximab in the 6 to 12 months prior to vaccination was associated with a failure to produce detectable COVID-19 antibodies, even after a third dose of the mRNA vaccines. In addition, some patients on Bruton tyrosine kinase (BTK) inhibitors also fail to make detectable COVID-19 antibodies, though individual results in BTK inhibitor patients were more variable.
Current cancer treatment guidelines do not call for pausing these treatments or delaying COVID-19 vaccination in blood cancer patients. The best path forward is for blood cancer patients to consult with their health care team and to get vaccinated and to continue practicing other important infection prevention procedures recommended by the CDC.
Rituximab, which uniformly blunted immune response in this study, is also used to treat patients with rheumatic diseases, including rheumatoid arthritis (RA) and lupus. Studies in rituximab-treated RA and lupus patients have also shown low seroconversion rates in these patients following the first two doses of mRNA COVID-19 vaccines.
According to Dr. Jeffrey Curtis, chair of the American College of Rheumatology (ACR) COVID Vaccine Guidance Task Force, “patients receiving rituximab for rheumatologic conditions should speak to their provider about the timing of vaccination in relation to their rituximab dosing. As this LLS study and other rheumatology studies have shown, patients receiving rituximab may be at particularly high risk to not mount an antibody response after vaccination against COVID and following other vaccines. It is for this reason that the ACR Task Force suggested that optimizing the timing of vaccination for rheumatology patients on rituximab may yield an improved response to primary vaccination and supplemental (booster) dosing.”
Current study expands on earlier findings from the LLS National Patient Registry
An earlier study of more than 1,400 blood cancer patients enrolled in the LLS National Patient Registry reported that about one in four fail to produce detectable antibodies after two doses of either Moderna or Pfizer mRNA COVID-19 vaccines. However, these rates vary by cancer type, with non-Hodgkin Lymphoma (NHL) and chronic lymphocytic leukemia (CLL) patients less likely to have detectable antibodies.
Patients with these forms of cancer have a diminished response to vaccination because NHL and CLL can deplete the body’s B-cells. The body needs healthy B cells to fight infections and also to mount an antibody response to the vaccines.
Among the 38 patients with B-cell derived malignancies (almost all had NHL or CLL) in this latest study who were seronegative after two vaccine doses, 21, or 55%, had detectable antibodies after the third dose while 17 patients (45%) remained seronegative (Figure below). The 11 patients in the study who had measurable antibodies after the first two doses all had increased antibody levels after the third dose. Even in this difficult-to-immunize population, the study shows the majority (32 of 49, 65%) have detectable antibodies.
“Antibody levels in our study ranged from 2.2 to over 2,500,” says Lee Greenberger Ph.D., LLS Chief Scientific Officer. “Antibodies tell us that a patient has responded to vaccination—and that is a positive finding, but vaccine experts are still working to determine exactly what antibody level is needed to protect against COVID-19 infection or its worst outcomes.”
Greenberger also says antibodies are just one part of the story. He and his colleagues, including Dr. Larry Saltzman, LLS Executive Research Director are working on another study assessing blood cancer patients’ T-cell response to vaccines. The immune system’s T-cells are primed by vaccination to create “killer cells,” which are the first line of defense against infections like COVID-19. They are also analyzing antibody data from hundreds of additional patients who have received a third vaccine dose. T-cell results and an update to the current study are both expected later this year.
Based on the results of this and the earlier LLS study, LLS estimates that at least 100,000 blood cancer patients in the U.S. will not have detectable antibodies following three doses of mRNA vaccines.
For more information, check out our COVID-19 Resource Page.
About the LLS National Patient Registry
The LLS National Patient Registry, a project of the Michael J. Garil Patient Data Collective, was created to honor the memory of Michael Garil, who was diagnosed with acute lymphoblastic leukemia in 1974 at the age of seven. His parents, Ethel and Bernard Garil have generously supported the creation of the LLS National Patient Registry to gather vital information from a large pool of people affected by blood cancers. The trial was conducted under IRB review with electronic consent of patients. Data were de-identified before being analyzed for vaccine results and all patient records are held in strict confidence.
Joining The LLS National Patient Registry today is safe and easy. Your participation will have a far-reaching impact on improving care for everyone with blood cancer. Visit LLS.org/Registry to learn more about the Registry and to sign up.
About The Leukemia & Lymphoma Society
The Leukemia & Lymphoma Society® (LLS) is a global leader in the fight against cancer. The LLS mission: Cure leukemia, lymphoma, Hodgkin's disease and myeloma. LLS funds lifesaving blood cancer research around the world, provides free information and support services, and is the voice for all blood cancer patients seeking access to quality, affordable, coordinated care.
Founded in 1949 and headquartered in Rye Brook, NY, LLS has regional offices throughout the United States and Canada. To learn more, visit www.LLS.org. Patients should contact the LLS Information Resource Center at (800) 955-4572, Monday through Friday, 9 a.m. to 9 p.m., ET.
For additional information visit lls.org/lls-newsnetwork. Follow us on Facebook, Twitter, and Instagram.
Contact:
Irene Tung
The Leukemia & Lymphoma Society
[email protected]
(718)-414-7910
References:
1. Bakouny Z, Hawley JE, Choueiri TK et al. (2020). COVID-19 and cancer: current challenges and perspectives. Cancer Cell. 38: 629-646. doi.org/10.1016/j.ccell.2020.09.018.
2. Vijenthera A, Gong IY, Fox TA et al (2020) Outcomes or patients with hematologic malignancies and COVID-19: a systemic review and meta-analysis of 3377 patients. Blood 136, 2881-2892. doi: 10.1182/blood.2020008824.